-
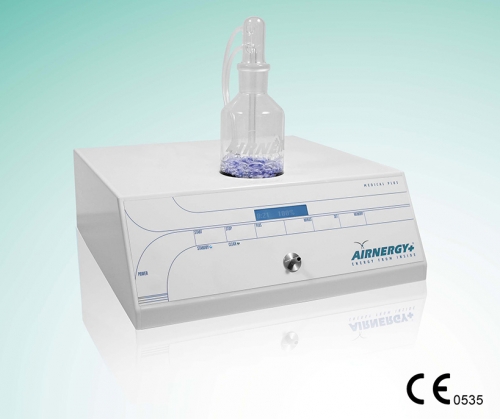 The Airnergy Medical Plus device has been approved as a medical device according to the basic requirements of the MDD (Medical Device Directive) 93/42/EEC. The device is used to supplement respiratory air (spirovitalisation), a process that can increase the energy available to the body from inhaled air.
The Airnergy Medical Plus device has been approved as a medical device according to the basic requirements of the MDD (Medical Device Directive) 93/42/EEC. The device is used to supplement respiratory air (spirovitalisation), a process that can increase the energy available to the body from inhaled air. -
 Avazzia BLUE Device, Y-175 Electrode, BEST Carry Case, lead wires, 2"x2" self-adhesive conductive gel pads - package of 4. Prescription Required. Soothing massage device that stimulates the body's natural responses to massage therapy for chronic or acute conditions. Designed for deeper therapy penetration. Prescription Required. The Avazzia Blue device has 2 modes, uses 2 AA 1.5V batteries, and has an accessory port for attaching lead wires and pads or electrode accessories.
Avazzia BLUE Device, Y-175 Electrode, BEST Carry Case, lead wires, 2"x2" self-adhesive conductive gel pads - package of 4. Prescription Required. Soothing massage device that stimulates the body's natural responses to massage therapy for chronic or acute conditions. Designed for deeper therapy penetration. Prescription Required. The Avazzia Blue device has 2 modes, uses 2 AA 1.5V batteries, and has an accessory port for attaching lead wires and pads or electrode accessories. *Medical Disclaimer
*Medical Disclaimer -
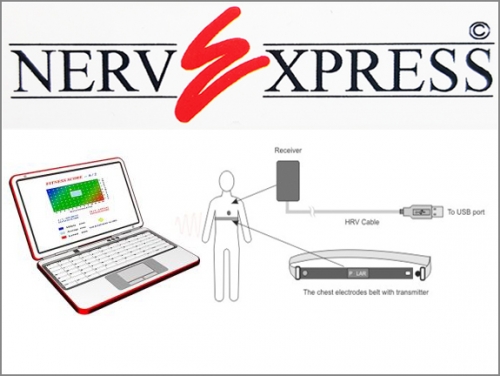 System requirements: IBM PC-compatible computer. Windows 8.0 or higher Operating System. Includes 1 Full Day Training Seminar or Webinar with additional 30 Days of telephone support. ( the most complete, practical, and immediately applicable clinical training available anywhere )
System requirements: IBM PC-compatible computer. Windows 8.0 or higher Operating System. Includes 1 Full Day Training Seminar or Webinar with additional 30 Days of telephone support. ( the most complete, practical, and immediately applicable clinical training available anywhere )

